How often have you needed to travel back to the pharmacy or doctor in order to refill that prescription of antibiotics? How about when you realized that you had forgotten to take your prescription dosage for that day- a simple mistake that yields potentially harmful effects? Well in the near-future, these commonplace issues may become a long-lost inconvenience with the embedding of implantable, drug-leaching microchips in our bodies. As far-fetched as it sounds, a study conducted by MicroCHIPS Inc. recently shown at the annual AAAS Conference in Vancouver, BC demonstrates just how modernistic this technology has become.

With a sample of eight Danish women suffering from osteoporosis, the microchip was implanted containing 20 doses of Teriparatide (brand name Forteo): a potent bone-building drug that normally requires daily injections to be effective. After one month’s trial, seven of the eight microchips delivered the osteoporosis drug in comparable doses to the daily injection, with no unwanted side effects. Even though the surgery for the implant must be done once a month with this size of implant, the subjects stated their preference for the device over the invasive injections normally needed by the osteoporosis drug.
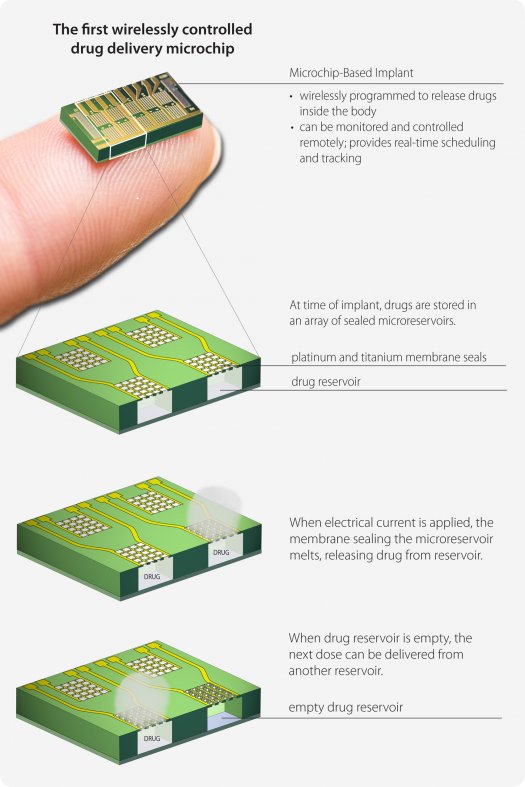
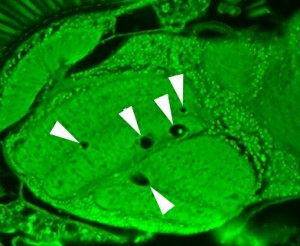
Visual representation of the drug-delivery implant in the osteoperosis patients. Courtesy of MicroCHIPS Inc.
As each of these devices contains nearly a month’s supply of a strong drug (in this case one for osteoporosis), a strong sealant and an intelligent way of communicating to the implant is needed to ensure an accidental leakage does not occur (left). The implant accomplishes this by locking up each of these wells with sealants of titanium and platinum, which melt away (at nontoxic levels) when an external frequency is sent by the physician. This FCC and FDA-approved signal observes the battery life, patient status, and confirmation that the proper dosage is being supplied. Now that the device has shown promise, researchers at the Massachusetts Institute of Technology (MIT) plan on testing the same mechanism in sizes of implants up to 365 wells, and with other drugs used to treat chronic illnesses.
Despite the device’s success, much research still needs to be conducted to determine its efficacy. For example, the body treats this implantation as foreign, and hence surrounds it with a layer of collagen that may be harmless, but could affect the delivery of the drug over time. Issues also arise with the device being electronic; not only could an electronic failure be fatal when this level of drugs is involved, but having an external radio source means that the device is also susceptible to hacking. MIT’s researchers plan on conducting future studies to address these concerns. If promising results continue, MicroCHIPS president David Farra predicts that the implant will be available for clinical use within only four years. Perhaps then, this drug-delivering microchip will join the ranks of the cochlear implant and pacemaker as our next big medical implant.
References
MicroCHIPS Inc. clinical trial: http://www.mchips.com/12_Feb_16_pr.html
Medicine.Net article: http://www.medicinenet.com/script/main/art.asp?articlekey=154949
Popular Science article: http://www.popsci.com/technology/article/2012-02/first-time-wirelessly-controlled-implantable-pharma-chip-delivers-drugs-inside-body