We are accustomed to thinking that science is conducted in a lab, but science is everywhere – even in your own home. Cooking is often viewed as a household skill, but it is actually a science, specifically, the science of ingredients and processes that change their tastes and forms to create a delicious product.
Eggs are a simple ingredient, and I’m sure many of us have experience cooking with it. Scrambled eggs, meringues, and Hollandaise sauce are all based on the humble egg, and yet yield such different results. How? It is all down to the science of the cooking methods!
Heat
Uncooked egg is composed of a clear, runny egg white and thick, yellow egg yolk. Egg whites are primarily water and protein, while yolks contain a higher percentage of proteins and considerably more lipids. Recall that proteins are composed of chains of amino acids. While the bonds linking individual amino acids are strong covalent bonds, the bonds holding the chains in its 3-dimensional structure are weaker hydrogen bonds. When the egg is heated, the heat denatures the proteins by breaking the weaker hydrogen bonds. This causes the proteins to unravel from their native configuration, leaving chains of unfolded protein. When these chains encounter one other, hydrogen bonds form between them at random, resulting in a network of interconnected proteins, which hardens the egg. Light can no longer penetrate through this mass of protein, and the egg white turns from clear to white. When the egg is heated for too long, the egg hardens too much and gives off a “rubbery” texture.
On a tangent: scientists recently devised a way to “un-scramble” an egg while investigating cancer research techniques. When cancer-associated proteins are produced in a lab, they often come out as a jumbled protein network akin to heated egg whites. This method of reversing denatured proteins may make cancer research more time and cost efficient.
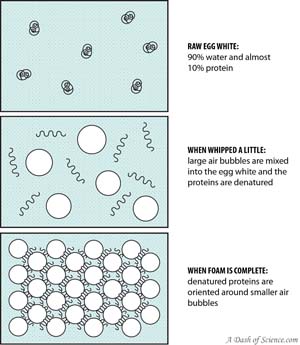
Beating
Meringues, soufflés, and tiramisu – all these light and fluffy textures come from beating egg whites. Beating egg whites adds air into the mixture, but the physical action also denatures the proteins, exposing hydrophobic and hydrophilic areas. As with heating, the denatured proteins cross-link to form a protein network, but with hydrophobic areas facing towards the air bubbles and hydrophilic areas facing away. The air bubbles are “locked” and incorporated into the network.
This does not work with yolks however, which contain lipids. The lipids interfere with the formation of the protein network, competing against proteins for a space to bind to. Recipes that call for egg foams will warn for careful separation of whites and yolks.
Emulsifier
Egg yolks have their own use as an emulsifier, combining oil and water mixtures that would otherwise separate. The hydrophobic/hydrophilic nature of amino acids in many yolk proteins (e.g. lecithin) attract water in some areas and oil in others, creating a thorough mixture of the two substances within the protein chains. Beating the mixture with a whisk further helps incorporate the liquids. This allows us to enjoy delicious creamy mayonnaise and Hollandaise sauces without them separating first!
Food for thought next time you’re pondering the scientific reasoning behind the steps in your recipe book!
– Peggy Hung