With summer just around the corner, it’s nearing that time of year where we start slapping on sunscreen to protect ourselves. After all, it’s well known that ultraviolet (UV) rays from the sun can damage our DNA. Despite this, UV light is still used for all sorts of applications – ranging from UV lamps at nail salons, to the hardening of resins.

A UV lamp in action. Source: cottonbro, Pexel.
The question remains: is it necessary to use UV light in these processes?
Good news: no!
Alternatives to UV light for similar processes have been recently identified by researchers. One such researcher is Taylor Wright, a graduate student at the University of British Columbia. In 2020, Wright developed a safe, low-cost method which uses harmless green LED lights instead of UV light. Wanting to learn more about this research, we sat down for an interview with Wright.
Into the details of cross-linking
The previously mentioned applications (UV lamps, resin hardening) are examples of a process called cross-linking. For an explanation of cross-linking, as well as a walkthrough of Wright’s cross-linking method of a material called polydimethylsiloxane (PDMS), please refer to the following video:
Okay… why should I care about this?
As we’ve previously said, frequent use of UV light in cross-linking processes poses a health risk to workers. By substituting with green LEDs, Wright’s newly developed method could help eliminate potential health complications in workers who deal with cross-linking processes.
But wait, there’s more! This research can also be applied to the biomedical field, through making antimicrobial fabrics. Turns out, by slightly altering the chemical properties of PDMS, we can give PDMS antimicrobial properties. By dipping a piece of fabric into liquid PDMS, then cross-linking to produce a solid PDMS coating, we can produce antimicrobial fabrics! The diagram below helps visualize the process.
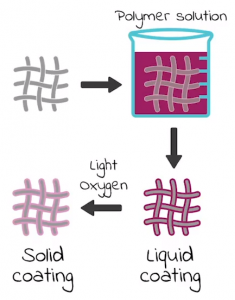
Step-by-step walkthrough of making the antimicrobial fabric. Source: Wright’s 3MT
This would have important implications for reducing the rate of infections in high-risk settings, such as hospitals. In the US alone, there are approximately 100,000 deaths resulting from infections acquired in a healthcare setting. Considering the COVID-19 pandemic that we’ve been in for a whole year now, this application has never been more relevant. Wright’s low-cost, simple, and safe method of producing antimicrobial fabrics could be crucial in reducing the impacts of any future pandemics.
Beyond the research
Although we learned much about his research during our interview with Wright, we were also able to get a glimpse into the person behind the science. To hear about his journey that resulted in the person he is today, listen to this week’s episode of Vancity Science, a podcast run by one of our members, Chie!
Audio sources: Blue Dot Sessions, Free To Use Sound – Royalty Free Sound Effects, Kyster, SnakeBarney, dobroide, KelliesKitchen, ZyryTSounds, ShadyDave, InspectorJ
Special thanks to Taylor Wright – we are excited to see further innovations by scientists with unique perspectives that can make the world a safer and better place!
– Sam Jung, Chie Nakayama, Madeline Filewych





