Grade level: Grade 4 Science
Topic: Energy Forms and Changes
Misconception: That energy is a “thing” in and of itself; it does not come in a variety of forms. That energy cannot be transferred from one object to another.
While students generally understand that adding heat warms a material and removing heat (“adding cold” i.e., ice) cools a material, the concepts of various forms of energy and energy transfer are much more abstract, and are likely to cause misconceptions and misunderstandings for students. The lesson I am outlining corresponds to the British Columbia fourth grade science curriculum (Big Idea: Energy can be transformed/Content: Energy has various forms; energy is conserved; devices that transform energy).
Objectives:
• Students will be able to demonstrate an understanding of different forms of energy.
• Students will be able to demonstrate how energy transfers from one object to another.
• Students will be able to explain the energy system they created in terms of energy input, energy output, and energy changes
Materials:
• Chart paper, Pens (x6)
• Computers to run PhET activity (requires Java Runtime Environment)
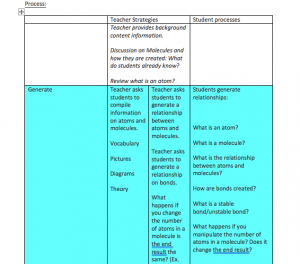
Step one: Generate ideas related to current concept(s)
Supporting inquiry: Students will be asked to respond to and develop hypotheses regarding the following questions:
• Question 1: What is energy input? What is energy output?
• Question 2: Does energy come in various forms?
• Question 3: Can energy be transferred from one object to another?
To complete this portion of the lesson, students will work in small groups (three to four students). Six stations will be set-up around the classroom with a piece of chart paper in the middle of the station (paper could also be hung on walls around classroom). Two stations will have question 1 written at the top of the chart paper, two stations will have question 2 written at the top of the chart paper, and two stations will have question 3 written at the top of the chart paper. Each group will have five minutes (time adjusted as necessary) at each station to brainstorm, discuss, and record ideas related to the question at the top of their chart paper. Groups will rotate three times so that each group will answer each of the three questions. At the end of the third rotation, each group will report out the ideas shared on the chart that they are currently at.
Step two: Hypothesize
Each group will be asked to create a hypothesis for each of the three questions. Their hypothesis can be based on their own ideas generated during the chart stations, or on the ideas of others. Students are welcome to walk around and look at the ideas generated at any of the chart paper stations to help them develop their hypotheses.
Step three (…to the computers!):
(Teachers may prefer students to work individually or in pairs at this point, depending on class composition, independence, and availability of computers)
Activity: PhET “Energy Forms and Changes” simulation “Intro”
<found at: https://phet.colorado.edu/en/simulation/legacy/energy-forms-and-changes>
Students will begin by exploring energy input, output and conservation, as well as thermal energy, by interacting with the “Intro” simulation. In this simulation, students experiment with heating and cooling iron, brick, and water, and are able to add or remove energy by heating (visual: fire) or cooling (visual: ice) the materials given. By clicking the “Energy Symbols” box, students are able to watch the transfer of energy as the temperature of the materials increases or decreases.
Step four (still on computers):
Activity: PhET “Energy Forms and Changes” simulation “Energy Systems”
Once students have completed the initial “Intro” to the “Energy Forms and Changes” simulation, students will click on the “Energy Systems” tab at the top left of the page to take them to the second simulation. This simulation allows students to see in more detail how energy is transferred and transformed as it moves between objects. Students build their own systems using a variety of energy sources, changers, and users, allowing students to opportunity to visually follow the transfer of energy throughout the system they have created.
Systems may be constructed using the following materials:
• wheel (turned by water, steam, bicycle), solar panel
• water tap, sunshine [with or without clouds], kettle, bicycle with rider
• container of water, regular light bulb, energy smart light bulb.
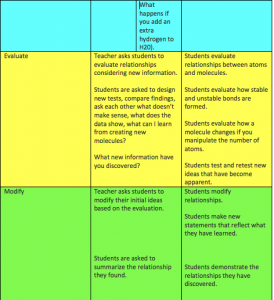
Step five: Re-evaluate ideas
Once students have completed both simulations, they will return to their hypothesis groups to re-evaluate their original hypotheses since gaining experience and knowledge while participating in the two simulations. The teacher will circulate to have each group explain how and why their original hypotheses changed after exploring “Energy Forms and Changes.”
A follow-up activity could be to have students complete a written response to answer the questions originally posed above (in step one), or to explain how their personal hypotheses changed throughout the course of the lesson. This would provide the teacher with individual feedback regarding understanding and possible continued misconceptions, as well as reconnect students to the original questions one more time.
Theory behind it:
Xiang and Passmore (2015) address the fact that the focus for science education has shifted “…from typical classroom practice that emphasizes the acquisition of content to a classroom in which students are active participants in making sense of the science they are learning” (p. 311). One of the leading principles discussed, behind this shift, is model-based inquiry, which can extend to include simulations. Through constructing, applying, and modifying models, students are given the opportunity to actively engage in the learning process, in addition to discussing, negotiating, and re-evaluating their conceptual models with peers (Xiang and Passmore, 2015). Finkelstein, Perkins, Adams, Kohl, & Podolefsky (2005) found that when the right learning environment was created, simulations could be equally effective, if not more effective, learning tools than traditional laboratory equipment “both in developing student facility with real equipment and at fostering student conceptual understanding” (p. 1-2). By integrating digital technology, not only can teachers access innovative and immersive learning environments for their students, but a number of factors can also be addressed, such as interest, motivation, and feasibility.
Srinivasan et al., (2006) highlight that “generally speaking, it is less expensive to develop a simulation than to provide real experience” (p. 137). While Srinivasan, et al. point out that this is especially clear in cases like cockpit simulators, this observation could be applied to many different science simulations, including the above PhET simulation used to teach fourth grade students about energy changes and transfer. While the PhET simulation is free (assuming school have access to computers and the internet), the time and cost associated with setting up a lab experience of the same depth would make the “real life” version of the simulations offered above unfeasible. In addition to providing an experience that would not be feasible without digital technology, novelty and interest, both addressed by Srinivasan et al. (2006) as motivational variables, are targeted in the simulations provided by PhET as well. Students are given the opportunity to explore science using a digital simulation which has the potential to increase interest and motivation as they actively engage with a simulated learning experience that would not have been possible in traditional elementary classroom settings.
References:
Energy forms and changes. (n.d.). Phet Interactive Simulations, University of Colorado. Retrieved from https://phet.colorado.edu/en/simulation/legacy/energy-forms-and-changes
Finkelstein, N.D., Perkins, K.K., Adams, W., Kohl, P., & Podolefsky, N. (2005). When learning about the real world is better done virtually: A study of substituting computer simulations for laboratory equipment. Physics Education Research, 1(1), 1-8.
Science 4: Big idea and content. (n.d.). British Columbia Ministry of Education. Retrieved from https://curriculum.gov.bc.ca/curriculum/science/4
Srinivasan, S., Perez, L. C., Palmer, R., Brooks, D., Wilson, K., & Fowler. D. (2006). Reality versus simulation. Journal of Science Education and Technology, 15(2), 137-141.
Xiang, L., & Passmore, C. (2015). A framework for model-based inquiry through agent-based programming. Journal of Science Education and Technology, 24(2-3), 311-329.